Teva's Fremanezumab Meets all Primary & Secondary Endpoints Across Both Monthly and Quarterly Dosing Regimens in Phase III Study in Episodic Migraine Prevention | Teva Sweden AB

Teva Announces Ministry of Health, Labour and Welfare Approves AJOVY® ( fremanezumab) Injection in Japan for the Preventive Treatment of Migraine in Adults
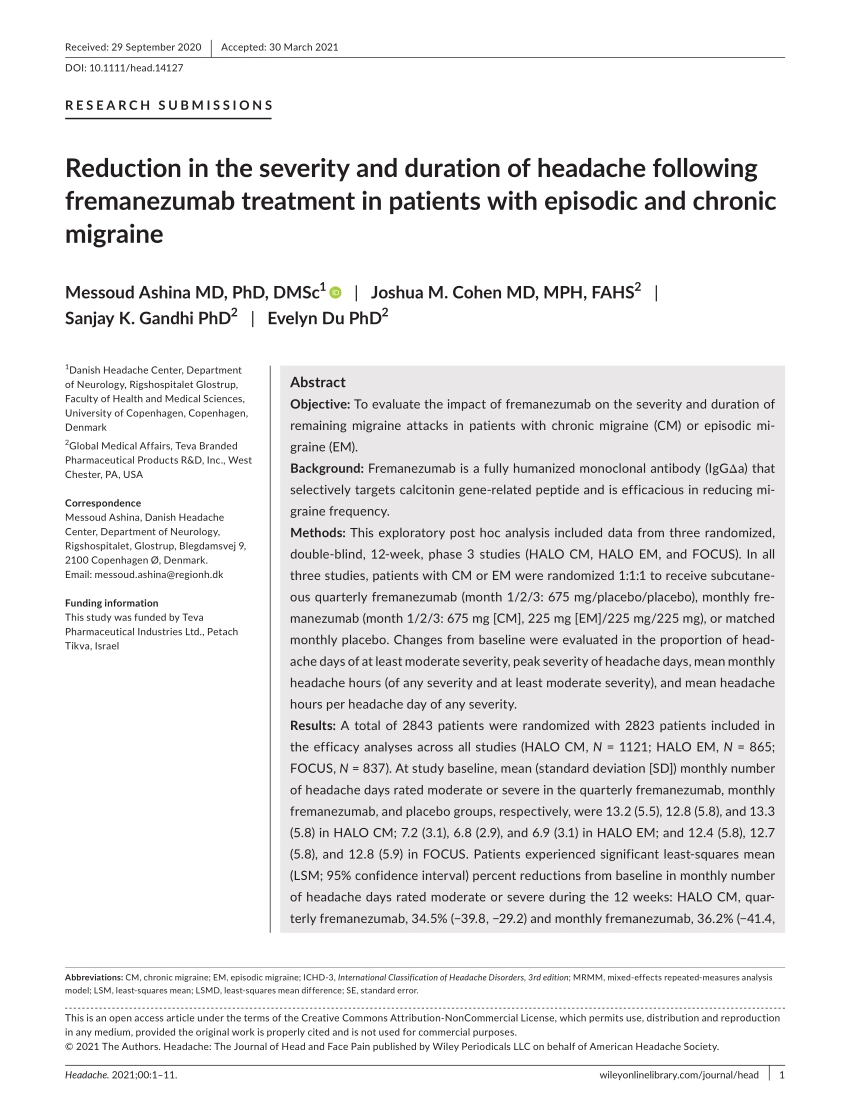
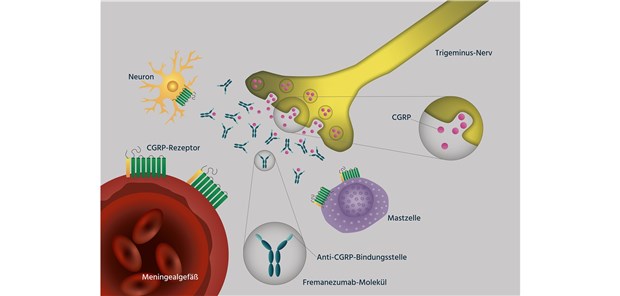
PDF) Reduction in the severity and duration of headache following fremanezumab treatment in patients with episodic and chronic migraine
Teva lanciert AJOVY zur Behandlung von Migräne mit dem YpsoMate Autoinjektor von Ypsomed - Ypsomed - Gruppe

Teva Canada Announces New Autoinjector for AJOVY® for the Preventive Treatment of Migraine in Adults | Business Wire

Orit Cohen-Barak's research works | Teva Pharmaceutical Industries Ltd., Petah Tikva (TEVA) and other places